A fast-aging fish revealed how kidneys grow old—and how a common drug slows the damage.
A new study published in Kidney International shows that medications known as SGLT2 inhibitors helped prevent age-related damage to kidney structure and function in the African turquoise killifish. This small vertebrate lives its entire life in just a few months, giving researchers a rare chance to observe aging unfold at high speed. The results help clarify the biological processes behind the strong kidney and heart protection these drugs provide in people, effects that go beyond their original role in controlling blood sugar.
The findings also establish the African turquoise killifish as a powerful new research model for understanding how organs age and for testing treatments that may help preserve organ function later in life.
A Fish That Recreates Human Kidney Aging in Months
The African turquoise killifish is one of the fastest aging vertebrates known, with a full lifespan of only four to six months. In this study, an international team of 13 researchers from MDI Biological Laboratory, Hannover Medical School, and Colby College found that the fish develops kidney changes over time that closely resemble those seen in aging humans.
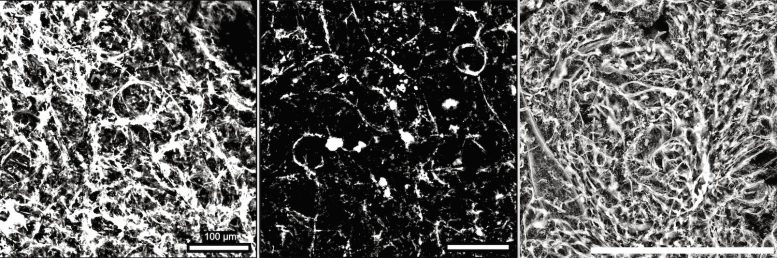
As the fish grew older, their kidneys lost tiny blood vessels, showed damage to the filtration barrier, developed higher levels of inflammation, and experienced disruptions in how kidney cells produce and manage energy. These changes are well-established features of kidney aging and disease in people.
Because the fish compresses decades of human-like kidney aging into a short period, researchers can follow the entire aging process and test interventions much faster than in longer-lived animals such as mice.
A Common Drug Reveals New Protective Effects
After confirming that the killifish accurately models kidney aging, the researchers examined the effects of sodium glucose cotransporter 2 (SGLT2) inhibitors. These drugs are widely prescribed to treat diabetes associated heart disease and chronic kidney disease.
“These drugs are already known to protect the heart and kidneys in patients with and without diabetes,” said Hermann Haller, M.D., senior author of the study and President of MDI Biological Laboratory. “What has been less clear is how they do so.”
Fish treated with SGLT2 inhibitors maintained healthier kidneys as they aged. Their kidneys preserved denser networks of capillaries, retained a stronger filtration barrier, and showed more stable energy production within kidney cells.
The treatment also helped maintain communication between different kidney cell types and reduced age-related inflammatory activity at the level of gene expression.
“Together, these upstream effects provide a biological explanation for clinical observations that the benefits of SGLT2 inhibitors often exceed what would be expected from glucose control alone,” Haller said. “They help explain why these drugs consistently reduce kidney and cardiovascular events across diverse patient populations.”
Protecting Blood Vessels and Cellular Energy
In untreated fish, a major driver of kidney decline was the gradual loss of capillaries, a process known as vascular rarefaction. As these small blood vessels disappeared, kidney cells shifted away from efficient mitochondria-based energy production and relied more heavily on less efficient backup pathways.
In contrast, kidneys from fish that received SGLT2 inhibitors retained healthier capillary networks and showed patterns of gene activity that more closely matched those of younger animals. These “youthful transcriptional profiles” were linked to improved energy metabolism and lower levels of inflammation.
Accelerating Aging Research With Human Impact
The study’s first author, Anastasia Paulmann, M.D., is a former postdoctoral researcher at MDI Bio Lab and also holds a clinical position at Hannover Medical School. She established and raised the new killifish colony at the Lab’s Kathryn W. Davis Center for Regenerative Biol0gy and Aging. Paulmann says the model offers a faster and more practical way to study aging with direct relevance to human health.
“Seeing these effects emerge so clearly in a rapid-aging model like our killifish was striking,” Paulmann said. “What impressed me most was how a seemingly simple drug influences so many interconnected systems within the kidney—from blood vessels and energy metabolism to inflammation and overall function.”
By allowing researchers to observe decades of kidney aging within months, the model creates a practical pipeline for testing how existing and experimental therapies affect organ resilience over time. This approach can help identify the most promising treatments before they move into human clinical trials.
The research team is now planning follow-up studies to explore whether SGLT2 inhibition can help repair kidney tissue after age-related damage has already occurred, and how treatment timing and duration shape long-term outcomes.
Reference: “Sodium-glucose co-transporter 2 inhibition improves age-dependent kidney microvascular rarefaction” by Anastasia Paulmann, Matthew D. Cox, Tom Boewer, Hannah M. Somers, Heath Fuqua, Ryan P. Seaman, Joel H. Graber, Anchal Mahajan, Cory P. Johnson, Laura L. Beverly-Staggs, Sonia Sandhi, Heiko Schenk and Hermann Haller, 23 December 2025, Kidney International.
DOI: 10.1016/j.kint.2025.12.011
This future work will be supported by expanded laboratory facilities at MDI Bio Lab as part of the institution’s MDI Bioscience initiative, which focuses on translating basic discoveries into strategies that improve human health.
This research was supported by the National Institutes of Health (P30GM154610, P20GM203423), the Morris Discovery Fund, the Scott R. McKenzie Foundation, and MDI Biological Laboratory.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
link




